Conduction of Heat – study notes
Conduction is a mode of transmission of heat where heat flows from the hot end to the cold end and particles of the medium simply oscillate but do not leave their place.
Conduction of Heat has the following characteristics:
- Medium is necessary for conduction.
- It is a slow process.
- The path of heat flow may be zig-zag.
- Conduction takes place in solids.
- The temperature of the medium increases through which heat flows.
- heat flows from the hot end to the cold end and
- particles of the medium simply oscillate but do not leave their place.
Conduction of Heat and related Terms
Variable and steady state
If the temperature of every cross-section of the rod goes on increasing, then the rod is said to exist in a variable state.
If the temperature of every cross-section of the rod remains constant and no part of the rod absorbs heat then the state of the rod is called steady state.
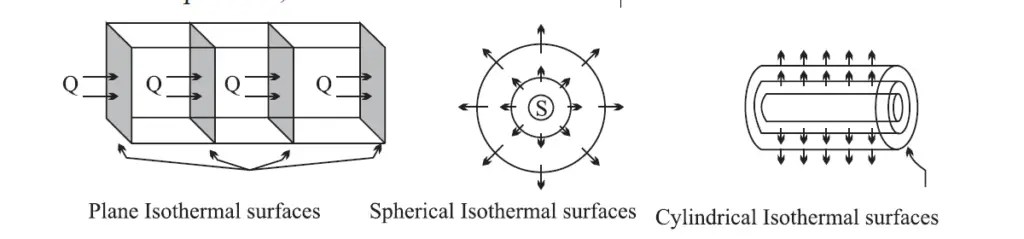
Isothermal surface
Any surface having its all points at the same temperature is called an isothermal surface.
Temperature Gradient
The rate of change of temperature with the distance between two isothermal surfaces is called temperature gradient.
Temperature gradient = [( θ – Δθ) – θ ] / Δx = – Δθ / Δx
The negative sign shows that temperature θ decreases as the distance x increases in the direction of heat flow.
Coefficient of thermal conductivity – formula
If L is the length of the rod, A the area of cross-section, and θ1 and θ2 are the temperatures of its two faces, then the amount of heat flowing from one face to the other face in time t is given by Q, where:
Q = KA ( θ2 – θ1 )t/L
Where K is the coefficient of thermal conductivity of the material of the rod. K is also called thermal conductivity.
Rate of flow of heat or Heat Current = H = Q/t
=> H = KA ( θ2 – θ1 )/L
The greater the value of K for a material, the more rapidly will it conduct heat.
K the coefficient of thermal conductivity – features
(i) Units = Js–1m–1k–1 or Wm–1k–1 (S.I.)
CGS unit = cal s–1 cm–1 ºC–1
(ii) Dimension : [MLT–3θ–1]
(iii) The magnitude of K depends only on the nature of the material.
(iv) For perfect conductors, K = ∞ and for perfect insulators, K = 0
(v) The thermal conductivity of pure metals decreases with the rise in temperature but for alloys, the thermal conductivity increases with the increase in temperature.
Thermal conductivities (K) of some material
| Material | Thermal conductivity (K) |
|---|---|
| Silver | 406 |
| Copper | 385 |
| Aluminium | 205 |
| Brass | 109 |
| Steel | 50.2 |
| Lead | 34.7 |
| Mercury | 8.3 |
| Glass | 0.8 |
| Ice | 1.6 |
| Wood | 0.12 |
| Water | 0.8 |
| Air | 0.024 |
| Hydrogen | 0.14 |
