What is Covalent Bonding? – study notes
Last updated on January 29th, 2022 at 03:00 pm
Covalent bonds are formed when atoms, rather than gaining or losing electrons, share them. Covalent bonds differ from ionic bonds in that there is no transfer of electrons involved. Electrical attraction between the nuclei of adjacent atoms and the shared electrons causes the atoms in a molecule to be held together. This force of attraction is called covalent bonding.
In this post, we will discuss this bonding in detail.
- What is covalent bonding?
- Examples of covalent bonds in nature
- Common properties of Covalent molecular substances
- Fuels are an important group of covalent molecular substances
- A theory for the structure of covalent molecular substances
- Covalent molecular element
- Covalent molecular compounds
- Tendency to achieve noble gas configuration
- chlorine atoms & covalent bonding
- Hydrogen – a special case of forming covalent bond
What is covalent bonding?
Most of the elements in the second and third periods of the periodic table need eight outer shell electrons for stability.
Nonmetallic atoms generally have high electronegativities. This means they can attract electrons easily. Except for hydrogen, nonmetals have four or more outer-shell electrons.
Covalent Bonding: When two nonmetallic atoms react together, both of them need to gain electrons in order to complete their outer shells. Since both cannot become negative ions, they share pairs of electrons to achieve eight electrons in the outer shell. This sharing of electron pairs between two non-metallic atoms produces a covalent bond. The atoms bond in order to reach a more stable state.
Examples of covalent bonds in nature
- Nitrogen and oxygen, are examples of covalent molecular elements.
- Drinking water regardless of its source is a covalent molecular compound.
- Rocks, sand, and minerals found in the solid part of the Earth contain substances such as quartz and diamond, which are classified as covalent network lattices.
- All these substances listed above are composed of non-metals only and behave very differently from the metallic and ionic substances. This different behavior is due to the unique structure that results when non-metal atoms are held together by a covalent bond. These bonds are formed when atoms, rather than gaining or losing electrons, share them. Covalent bonds differ from ionic bonds in that there is no transfer of electrons involved.
Common properties of Covalent molecular substances
- The forces of attraction between the particles are very weak.
- No charged particles are free to move through the substance.
Fuels are an important group of covalent molecular substances
Fuels are an important group of covalent molecular substances.
- The gas that is piped to homes is mainly methane (CH4).
- Bottled (LPG) gas used for barbecues and camping is a mixture of propane (C3H8) and butane (C4H10).
- Octane (C8H18) is a major component of petrol.
- Methanol (CH3OH) and ethanol (C2H5OH) are fuels used in high-performance racing cars.
These substances all share the ability to ignite easily and burn in oxygen to release large amounts of heat. These compounds are all gases or liquids at room temperature and pressure. This means that they have low boiling and melting temperatures. They are also poor conductors of electricity.
A theory for the structure of covalent molecular substances
The major features of the current theory of the structure of covalent molecular substances are as follows:
• The basic units of covalent molecular substances are groups of atoms called molecules. All molecules within a substance are identical.
• Adjacent atoms within a molecule share electrons in order to achieve a full outer shell.
• Electrical attraction between the nuclei of adjacent atoms and the shared electrons causes the atoms in a molecule to be held together. This force of attraction is called covalent bonding.
• The overall charge on each molecule is zero and so adjacent molecules are held together by weak intermolecular forces.
Covalent molecular element
A covalent molecular element is made up of identical atoms held together by covalent bonds. Such elements may be found in various arrangements, including diatomic molecules. These molecules are described as discrete, as each molecule is separate and distinct from the others.
Diatomic molecules are composed of two atoms of the same element. For example, the covalent molecular element hydrogen exists as a gas made up of discrete diatomic molecules. As shown, each molecule is composed of two hydrogen atoms. This is represented by the symbol H2.
Hydrogen is one of several nonmetallic elements that are made up of diatomic molecules.
Other molecular elements include oxygen (O2), nitrogen (N2), phosphorus (P4), fluorine (F2), and sulfur (S8).
The noble gases, helium, neon, argon, and krypton, all exist as discrete atoms.
Since they have full outer shells, they are unreactive and few of their compounds are known.
Covalent molecular compounds
In addition to these elements, a large number of compounds also exist as molecules.
In a molecular compound, atoms of different elements share electrons with each other. These compounds are often called covalent molecular compounds because they contain covalent bonds. Water, carbon dioxide, and methane are examples of these compounds.
Tendency to achieve noble gas configuration
Atoms of elements with a full outer shell, such as the noble gases helium, neon, and argon, are particularly stable.
Other atoms tend to achieve noble gas configuration – that is, a full outer shell – by losing or gaining electrons to form ionic bonds, or sharing electrons to form covalent bonds.
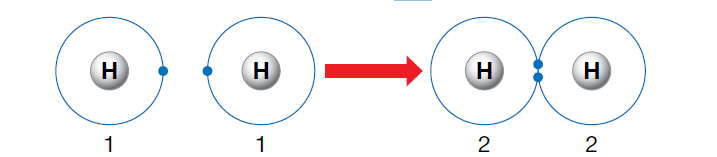
The hydrogen atom needs two electrons in its outer shell for stability.
chlorine atoms & covalent bonding
To better understand this process of covalent bonding, we can use electron shell diagrams.
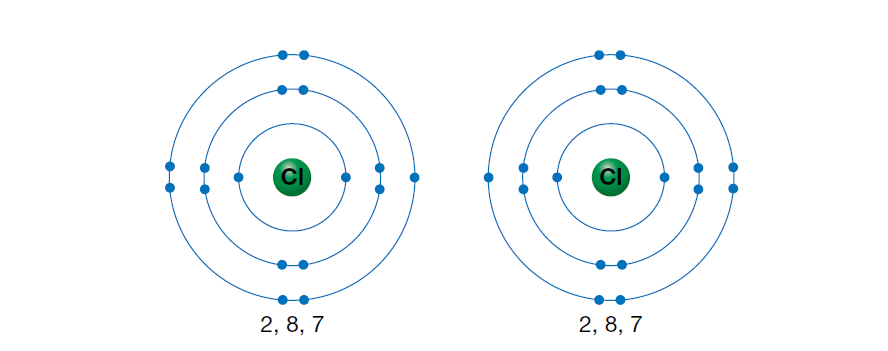
Consider the two chlorine atoms shown in the figure below.
Chlorine, as well as all members of group 17, exists in nature as diatomic molecules (two atoms covalently bonded together). Each chlorine atom has seven valence electrons. In order to attain a noble gas configuration (a full outer shell of eight electrons), each chlorine atom needs one electron.
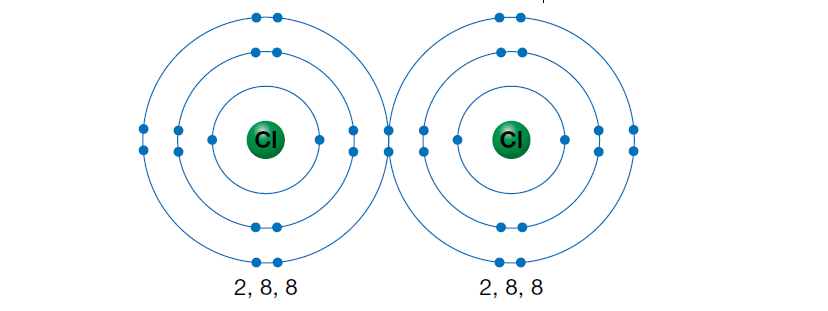
By sharing the single, unpaired electrons, both atoms can complete their outer shells, as shown in the figure below. A covalent bond results from the simultaneous attraction of the two positive chlorine nuclei to the same shared pair of electrons. This bond holds the two atoms together. A molecule of chlorine forms and is represented by the symbol Cl2.
Hydrogen – a special case of forming covalent bond
The hydrogen atom is an exception. This is because each of two hydrogen atoms contains one electron in its first (and outer) shell. They both need another electron to attain the stable electron configuration, 2, of the nearest noble gas, helium. This is achieved by each atom gaining a share in the electron of the other and forming a covalent bond, as shown in the figure below. In this way, the diatomic hydrogen molecule, H2, is formed.