Important formulas related to Atomic Structure
In this post, you will find a bunch of important formulas related to atomic structure.
formulas related to atomic number & mass number
(a) Atomic number (Z) = number of electrons = number of protons
(b) Mass number (A) = number of protons + number of neutrons = atomic number + number of neutrons
(c) Number of protons = atomic number = mass number – number of neutrons
(d) Number of electrons = atomic number = mass number – number of neutrons
(e) Number of neutrons = mass number – atomic number
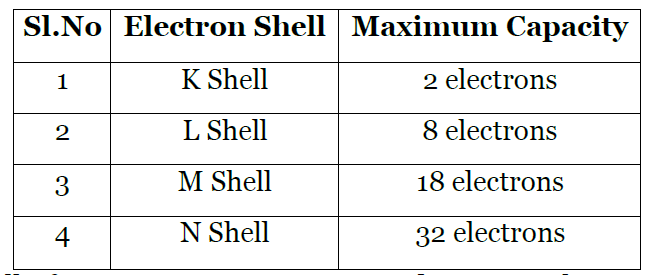
Bohr and Bury Scheme – Maximum number of electrons in energy levels
Bohr and Bury Scheme – Maximum number of electrons in energy levels
(i) Maximum number of electrons that can be accommodated in a shell is given by 2n2 where n = shell number
(ii) For 1st energy level (K shell), n = 1
Maximum number of electrons in 1st energy level = 2n2 = 2 x (1)2 = 2
(iii) For 2nd energy level (L shell), n = 2
Maximum number of electrons in the 2nd energy level = 2n2 = 2 x (2)2 = 2 x 4 = 8
(iv) For 3rd energy level (M shell), n = 3
Maximum number of electrons in the 3rd energy level = 2n2 = 2 x (3)2= 2 x 9 = 18
(v) For 4th energy level (N shell), n = 4
Maximum number of electrons in the 4th energy level = 2n2 = 2 x (4)2 = 2 x 16 = 32
The outermost shell of an atom cannot accommodate more than 8 electrons, even if it has the capacity to accommodate more electrons. This is a very important rule and is also called the Octet rule. The presence of 8 electrons in the outermost shell makes the atom very stable.
