Structure of Water molecule – revision notes
In a water molecule, an oxygen atom shares a pair of electrons with each of two hydrogen atoms with polar covalent bonds. Oxygen has six outer electrons and needs two more to satisfy the octet rule, achieving the noble gas structure of eight.
Each hydrogen atom needs one more electron to fill its outer orbital with two. Therefore, one oxygen atom bonds with two hydrogen atoms, forming H2O.
Both oxygen and hydrogen are more stable with the outer orbital configuration of the noble gases (neon and helium in this case).
electron sharing in water molecule | Polar molecule & dipole
Electrons are shared in a water molecule but not equally.
Oxygen, with its eight positive protons, has a greater attraction for the shared electrons than do either of the hydrogens with a single proton.
Therefore, the shared electrons spend more time around the oxygen part of the molecule than they do around the hydrogen part. This results in the oxygen end of the molecule being more negative than the hydrogen end. When electrons in a covalent bond are not equally shared, the molecule is said to be polar. A polar molecule has a dipole (di=two; pole=side or end), meaning it has a positive end and a negative end.
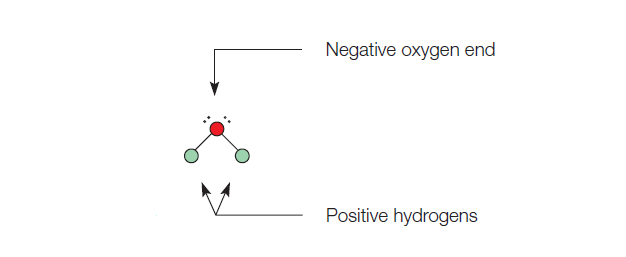
model of water molecule showing its polar nature (with diagram)
A water molecule has a negative center at the oxygen end and a positive center at the hydrogen end. The positive charges on the hydrogen ends are separated, giving the molecule a bent rather than straight-line arrangement. Figure 1 shows a model of a water molecule showing its polar nature.
Polar molecules and hydrogen bonding
It is the polar structure of the water molecule that is responsible for many of the unique properties of water.
Polar molecules of any substance have attractions between the positive end of a molecule and the negative end of another molecule. When the polar molecule has hydrogen at one end and fluorine, oxygen, or nitrogen at the other, the attractions are strong enough to make a type of bonding called hydrogen bonding.
Hydrogen bonding is a strong bond that occurs between the hydrogen end of a molecule and the fluorine, oxygen or nitrogen end of similar molecules.
A better name for this would be a hydrogen-fluorine bond, a hydrogen-oxygen bond, or a hydrogen-nitrogen bond. However, for brevity, the second part of the bond is not named, and all the hydrogen-something bonds are simply known as hydrogen bonds.
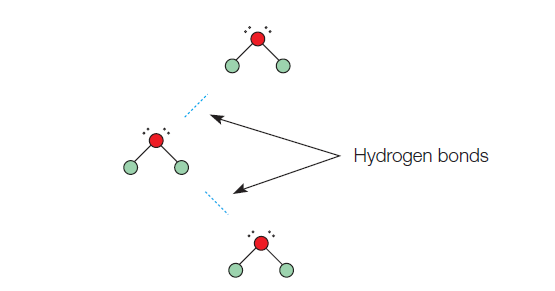
The dotted line between the hydrogen and oxygen molecules in Figure 2 represents a hydrogen bond. A dotted line is used to represent a bond that is not as strong as the bond represented by the solid line of a covalent compound.
Hydrogen bonding accounts for the physical properties of water, including its unusual density changes with changes in temperature.
