Atomic Structure – shell model, based on the Bohr model
In this post, various aspects of the shell model of the atomic structure (based on the Bohr model) have been discussed.
- Electron Orbits
- electron as a spherical negative-charge cloud
- electrons in shells and subshells
- Labels of shells & subshells | principal and orbital angular momentum quantum numbers
- Carbon atom and subshells | electronic configuration of Carbon
- electrons in the outer subshells
- valence electron
- stable subshell
- Energy of electrons in shells & subshells
- ionization energy of the atom
- Electron affinity
Electron Orbits
There are as many orbiting electrons as there are protons in the nucleus. The radii of the electron orbits are assumed to be quite larger than the size of the nucleus. In other words, the electrons are assumed to be orbiting the nucleus at very large distances compared to the size of the nucleus. An important assumption in the Bohr model is that only certain orbits with fixed radii are stable around the nucleus. For example, the closest orbit of the electron in the hydrogen atom can only have a radius of 0.053 nm.
electron as a spherical negative-charge cloud
Since the electron is constantly moving around an orbit with a given radius, over a long time period (perhaps ∼10−12 seconds on the atomic time scale), the electron would appear as a spherical negative-charge cloud around the nucleus and not as a single dot representing a finite particle. We can therefore view the electron as a charge contained within a spherical shell of a given radius.
electrons in shells and subshells
Due to the requirement of stable orbits, the electrons, therefore, do not randomly occupy the whole region around the nucleus. Instead, they occupy various well-defined spherical regions. They are distributed in various shells and subshells within the shells, obeying certain occupation (or seating) rules. The example for the carbon atom is shown in Figure 1.1.
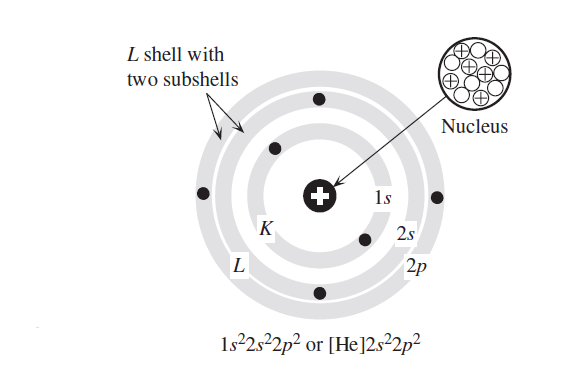
Labels of shells & subshells | principal and orbital angular momentum quantum numbers
The shells and subshells that define the whereabouts of the electrons are labeled using two sets of integers, n, and ℓ. These integers are called the principal and orbital angular momentum quantum numbers, respectively.
The integers n and ℓ have the values
n = 1, 2, 3, . . . , and ℓ = 0, 1, 2, . . . , n − 1, and ℓ < n.
For each choice of n, there are n values of ℓ, so higher-order shells contain more subshells.
The shells corresponding to n = 1, 2, 3, 4, . . . are labeled by the capital letters K, L, M, N, . . . , and the subshells denoted by ℓ = 0, 1, 2, 3, . . . are labeled s, p, d, f . . . .
The subshell with ℓ = 1 in the n = 2 shell is thus labeled the 2p subshell, based on the standard notation nℓ.
There is a definite rule to filling up the subshells with electrons; we cannot simply put all the electrons in one subshell. The number of electrons a given subshell can take is fixed by nature to be 2(2ℓ + 1). For the s subshell (ℓ = 0), there are two electrons, whereas, for the p subshell, there are six electrons, and so on.
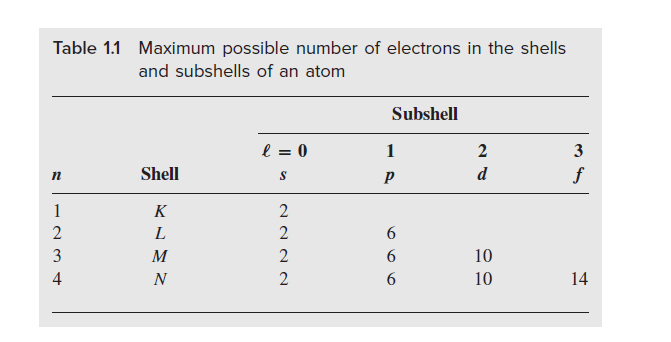
Table 1.1 summarizes the most number of electrons that can be put into various subshells and shells of an atom. Obviously, the larger the shell, the more electrons it can take, simply because it contains more subshells. The shells and subshells are filled starting with those closest to the nucleus.
Carbon atom and subshells | electronic configuration of Carbon
The number of electrons in a subshell is indicated by a superscript on the subshell symbol, so the electronic structure, or configuration, of the carbon atom (atomic number 6) shown in Figure 1.1 becomes 1s22s22p2.
electronic configuration of Carbon in terms of Inert element (He)
The K shell has only one subshell, which is filled with two electrons. This is the structure of the inert element He.
We can therefore write the electronic configuration more simply as [He]2s22p2. The general rule is to put the nearest previous inert element, in this case, He, in square brackets and write the subshells thereafter.
electrons in the outer subshells
The electrons occupying the outer subshells are the farthest away from the nucleus and have the most important role in atomic interactions, as in chemical reactions, because these electrons are the first to interact with outer electrons on neighboring atoms.
valence electron
The outermost electrons are called valence electrons and they determine the valency of the atom. Figure 1.1 shows that carbon has four valence electrons in the L shell.
stable subshell
When a subshell is full of electrons, it cannot accept any more electrons and it is said to have acquired a stable configuration. This is the case with the inert elements at the right-hand side of the Periodic Table, all of which have completely filled subshells and are rarely involved in chemical reactions. The majority of such elements are gases in as much as the atoms do not bond together easily to form a liquid or solid. They are sometimes used to provide an inert atmosphere instead of air for certain reactive materials.
Energy of electrons in shells & subshells
An electron that is closer to the nucleus has lower energy.
The electrons nearer the nucleus are more closely bound and have higher binding energies. The 1s22s1 configuration of electrons corresponds to the lowest energy structure for Li and, at the same time, obeys the occupation rules for the subshells.
If the 2s electron is somehow excited to another outer subshell, the energy of the atom increases, and the atom is said to be excited.
ionization energy of the atom
The smallest energy required to remove a single electron from a neutral atom and thereby create a positive ion (cation) and an isolated electron is defined as the ionization energy of the atom.
The Na atom has only a single valence electron in its outer shell, which is the easiest to remove. The energy required to remove this electron is 5.1 electron volts (eV), which is the Na atom’s ionization energy.
Electron affinity
The electron affinity represents the energy that is needed or released when we add an electron to a neutral atom to create a negative ion (anion).
Notice that the ionization term implies the generation of a positive ion, whereas the electron affinity implies that we have created a negative ion. Certain atoms, notably the halogens (such as F, Cl, Br, and I), can actually attract an electron to form a negative ion. Their electron affinities are negative.
When we place an electron into a Cl atom, we find that energy of 3.6 eV is released. The Cl− ion has lower energy than the Cl atom, which means that it is energetically favorable to form a Cl− ion by introducing an electron into the Cl atom.
Reference: PRINCIPLES OF ELECTRONIC MATERIALS AND DEVICES by S. O. Kasap, University of Saskatchewan, Canada
