Heat Engines
Last updated on May 16th, 2022 at 09:02 pm
This post talks of an important topic of the Thermodynamics chapter of Physics. We talk here about Heat Engines, their definition, examples of heat engines, internal combustion engines, internal combustion process, formulas and diagrams of heat engine, efficiency formulas of heat engines, etc.
- Heat Engine – definition
- Steam engine is a heat engine – explain
- Efficiency of heat engine can't be 100%
- efficiency of Internal combustion engine
- Internal combustion process
- Heat engine – in a diagram
- Second Law of Thermodynamics and Heat engine – in a diagram
- Heat Engine Formula
- Formulas of the efficiency of a heat engine
Heat Engine – definition
Any device that uses heat (thermal) energy to produce mechanical work is called a heat engine.
A heat engine is a device that converts heat into work in a cyclical process. That is, the engine periodically returns to its starting point.
A steam engine is a heat engine. Gasoline or diesel power automobile engines are all heat engines where the pistons return to their starting positions.
Steam engine is a heat engine – explain
One of the first heat engines was the steam engine. Let’s see how a steam engine used to work as a heat engine.
- Wood or coal was burned in a steam engine, and the thermal energy released by the fuel was transferred to water and produced steam.
- The steam was then directed into a cylinder with a movable piston which turned a crank of some sort that was attached to another mechanical device that turned a paddle wheel.
- Thus, work was performed.
- But plenty of thermal energy was also transmitted to the environment (the engine room on a steamship gets pretty hot!).
Efficiency of heat engine can’t be 100%
Using thermal energy to produce mechanical work led to the question of efficiency: Efficiency is the measure of how much of the energy input to a system can be used to produce a given amount of work.
It is never the case that all of the thermal energy can be converted to mechanical energy.
Here is another way of stating the second law of thermodynamics: no heat engine can be 100% efficient.
There is a simple example to appreciate this fact. If we ever try to feel the hood of an automobile after it’s been running for a while (to do this carefully) we’ll notice that the hood is very hot. The engine has grown hot while performing work and heated the hood of the car. The heat engine in this case is the internal combustion engine.
efficiency of Internal combustion engine
The internal combustion engine is not very efficient.
About two-thirds of the energy of the gasoline used to power an automobile is converted to heat. That is why automobiles have cooling systems composed of radiators, water pumps, and fans. As the water circulates through the system, heat is transferred from the hot engine to the cooler water. The water is then cooled by the radiator fan (and the “wind” through the grills of the car).
During the internal combustion process, some of the thermal energy produced by burning gasoline is used to perform work (to move the car!). But a good deal of thermal energy is used to heat the engine and the exhaust gases which do not perform work. The second law of thermodynamics tells us that only some of the chemical energy of gasoline is available to perform work.
Internal combustion process
Briefly, the internal combustion process is as follows.
- (Intake) Gasoline and air are mixed together in a cylinder that expands against a piston.
- (Compression) A crankshaft then moves the piston upward compressing the mixture.
- (Ignition) At the instant of maximum compression a spark plug “fires” (it releases an electric spark into the mixture) igniting the gasoline – air mixture and rapidly increasing the temperature in the cylinder.
- (Expansion—the power stroke) The hot mixture QH expands rapidly.
- (Exhaust) The exhaust gases QL are ejected at a lower temperature as the piston and the process repeats.
Heat engine – in a diagram
Figure 1 below explains how the typical heat engine works.
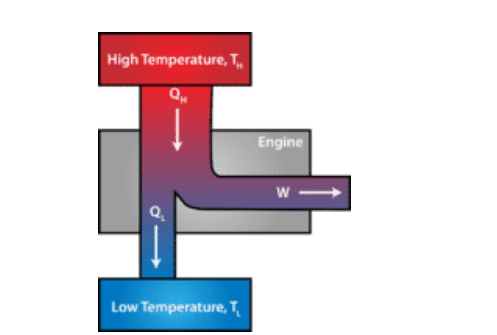
Figure 1 explanation:
The quantity QH is the initial thermal energy transferred to the system and W is the work done by the system. (In the case of the internal combustion process, QH is the heat produced when the spark ignites the gasoline-air mixture.)
The remaining thermal energy QL leaves the engine at a lower temperature.
Second Law of Thermodynamics and Heat engine – in a diagram
The Second Law can be shown in the diagram below where the input heat is taken from a “reservoir” that maintains a constant high temperature and the output goes to a second reservoir at a constant low temperature.
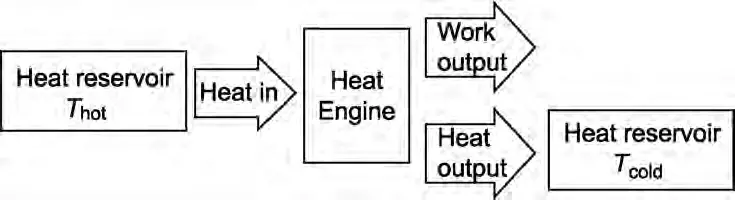
For example, a steam turbine gets its heat from high-temperature steam and puts its output heat into a much colder lake or river.
Heat Engine Formula
Let QH be the initial energy of the system, QL the energy of the spent gases, and W the work output.
Conservation of energy states Energyinitial = Energyfinal
=> QH = QL+W ………….. (1)
=> W = QH – QL ………..(2)
Formulas of the efficiency of a heat engine
The % efficiency η of a heat engine is defined as:
- η =(W/QH)×100% where QH is the initial thermal energy transferred to the system and W is the work done by the system.
- η =(QH-QL)/QH × 100%
- η =(1 – QL/QH) × 100%
- The % efficiency of any heat engine can also be expressed as η=(W/Qin) x 100%, where Qin is the initial thermal energy transferred to the system and W is the work done by the system.
- An ideal engine, the most efficient theoretically possible, is called a Carnot Engine. Carnot was able to show that at maximum efficiency the high QH and low QL energy transfers were proportional to the high TH and low TL temperatures under which the engine operated.
- We can therefore write e=(1 − (TL / TH))×100% where T is in kelvins, where
TL = the temperature of the exhaust environment (Tcold) and
TH = the temperature of the heat input ( Thot ), in Kelvins
- In a Carnot engine heat is input and exhausted in isothermal cycles, and the % efficiency of a Carnot (ideal) heat engine is
η=[1− (Tcold / Thot)] x 100%
Read about: Carnot Engine
