Excess Pressure Across a Curved Liquid Surface (surface tension notes)
In this post, we will cover an important topic of the Surface Tension chapter. And this topic is Excess Pressure Across a Curved Liquid Surface.
What is excess pressure across the liquid surface
In general, a liquid surface may be a plane, concave or convex. Due to the shape of the liquid surface and the force of surface tension, in general, the pressure across the two sides of the film is not the same. The difference is known as the excess pressure across the liquid surface.
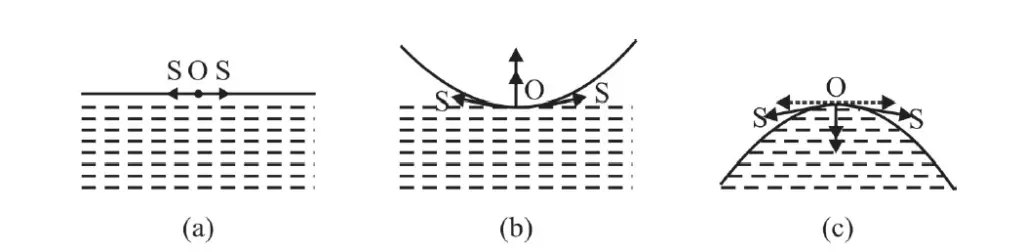
Analyzing 3 types of surface of liquid
Fig. (a) shows a molecule O on the plane liquid surface. The force of surface tension S, on O as shown in Fig a. The pressure across the two sides of the film is the same; i.e. there is no pressure difference across the two sides of a plane surface.
Fig. (b) shows a concave surface. The force of surface tension on molecule at O is shown in Fig. (b). Resolving S into horizontal and vertical components; there is a net outward force. Therefore pressure above the liquid surface is more than pressure below it. The pressure on the concave side is more.
Fig. (c) shows a convex surface. There is a net inward force on molecule O. In other words, pressure on the lower surface is more than on its upper surface. There is a pressure difference across the surface.
We can conclude that a curved surface of a liquid will be in equilibrium if there is excess pressure on the concave side of a curved surface.
