Surface Tension & Surface Energy – MCQ worksheets
In this post, we are presenting a few sets of MCQ worksheets with questions from the Surface Tension chapter of Physics. These sets include questions on Cohesive force and adhesive force, surface tension, and surface energy.
And here is the link to the next part of this surface tension MCQ set. MCQ on excess pressure & angle of contact.
Problems based on Cohesive and adhesive force
1 ) Mercury does not wet glass, wood, or iron because [MP PMT 1995; MP PET 1997]
(a) Cohesive force is less than adhesive force (b) Cohesive force is greater than the adhesive force
(c) Angle of contact is less than 90 degrees (d) Cohesive force is equal to the adhesive force
2 ) The force of cohesion is [CPMT 1996]
(a) Maximum in solids (b) Maximum in liquid (c) Same in different matters (d) Maximum in gases
3 ) What enables us to write on the blackboard with chalk
(a) Gravity (b) Cohesion (c) Adhesion (d) None of the above
4 ) Intermolecular forces decrease rapidly as the distance between the molecules increases and do so much more
(a) Slowly than demanded by the inverse square law of the distance
(b) Rapidly than anticipated through the inverse square law of the distance
(c) According to the inverse square law
(d) It actually remains the same for all the distances
Problems based on Surface tension
5 ) The spherical shape of rain-drop is due to
[CPMT 1976, 90; CPMT 2001; NCERT 1982; AIIMS 1998; MHCET 2000; DCE 1999; AFMC 1999, 2001]
(a) Density of the liquid (b) Surface tension (c) Atmospheric pressure (d) Gravity
6 ) At which of the following temperatures, the value of surface tension of water is minimum [MP PMT/PET 1998]
(a) 4o C (b) 25o C(c) 50o C(d) 75o C
7) Force necessary to pull a circular plate of 5cm radius from water surface for which surface tension is 75 dynes/cm, is [MP PMT 1991]
(a) 30 dynes (b) 60 dynes (c) 750 dynes (d) 750 Pi dynes
8 ) A square frame of side L is dipped in a liquid. On taking it out, a membrane is formed. If the surface tension of the liquid is T, the force acting on the frame will be [MP PMT 1990]
(a) 2TL (b) 4TL (c) 8TL (d) 10TL
9 ) Ball pen and fountain pen depend respectively upon the principle of
(a) Surface tension and viscosity (b) Surface tension and gravity
(c) Gravitation and surface tension (d) Surface tension and surface tension
10 ) Which graph represents the variation of surface tension with temperature over small temperature ranges for water
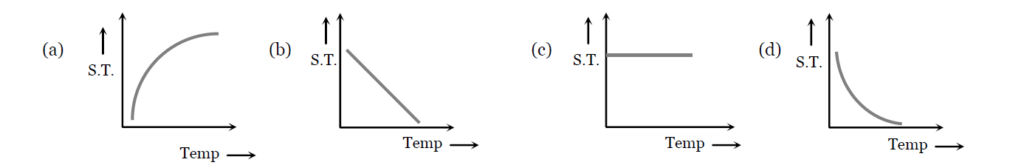
11 ) The material of a wire has a density of 1.4 g per cm3. If it is not wetted by a liquid of surface tension 44 dyne per cm, then the maximum radius of the wire which can float on the surface of the liquid is
(a)1/7 cm (b) 0.7 cm (c) 10/14 cm (d)10/28 cm
12 ) A water drop of 0.05 cm3 is squeezed between two glass plates and spreads into an area of 40cm2. If the surface tension of water is 70 dyne/cm then the normal force required to separate the glass plates from each other will be
(a) 90 N (b) 45 N (c) 22.5 N (d) 450 N
13 ) The main difference between a stretched membrane and the liquid surface is
(a) The liquid surface has a tendency to contract but the stretched membrane does not
(b) The surface tension does not depend on the area but on the tension of the stretched membrane does
(c) The surface tension increases with increases in area
(d) Surface tension increases irregularly with temperature
14 ) On bisecting a soap bubble along a diameter, the force due to surface tension on any of its half parts will be
(a) 4 π R T (b)4 π R/T (c) T/(4 π R) (d) 2T/R
15) The addition of soap changes the surface tension of water to T1 and that of sugar changes it to T2. Then
(a) T1=T2 (b) T1>T2 (c) T1<T2 (d) Can’t be predicted
16 ) A hollow disc of aluminum whose external and internal radii are R and r respectively is floating on the surface of a liquid whose surface tension is T. The maximum weight of the disc can be
(a) 2π (R + r) T (b) 2π (R – r) T (c) 4π(R + r) T (d) 4π(R – r) T
Problems based on Surface energy
17 ) 8000 identical water drops are combined to form a big drop. Then the ratio of the final surface energy to the initial surface energy of all the drops together is
(a) 1 : 10 (b) 1 : 15 (c) 1 : 20 (d) 1 : 25
18 ) 8 mercury drops coalesce to form one mercury drop, the energy changes by a factor of [DCE 2000]
(a) 1 (b) 2 (c) 4 (d) 6
19) Which of the following statements are true in the case when two water drops coalesce and make a bigger drop [Roorkee 1999]
(a) Energy is released
(b) Energy is absorbed
(c) The surface area of the bigger drop is greater than the sum of the surface areas of both the drops
(d) The surface area of the bigger drop is smaller than the sum of the surface areas of both the drops
20 ) An oil drop of radius 1cm is sprayed into 1000 small equal drops of the same radius. If the surface tension of oil drop is 50 dyne/cm then the work done is [RPET 1990]
(a) 18 π ergs (b) 180 π ergs (c) 1800 π ergs (d) 18000 π ergs
21 ) If work W is done in blowing a bubble of radius R from a soap solution, then the work done in blowing a bubble of radius 2R from the same solution is [MP PET 1990]
(a) W/2 (b) 2W (c) 4W (d) 7W/3
22 ) A liquid drop of radius R is broken up into N small droplets. The work done is proportional to
(a) N (b) N(2/3) (c) N(1/3) (d) N0
23 ) The work done in increasing the volume of a soap bubble of radius R and surface tension T by 700% will be
(a) 8 π R2 T(b) 24 π R2 T (c) 48 π R2 T (d) 8 π R2 T2 /3
24 ) 1000 drops of water all of the same size join together to form a single drop and the energy released raises the temperature of the drop. Given that T is the surface tension of water, r is the radius of each small drop, ρ is the density of the liquid, J is the mechanical equivalent of heat. What is the rise in the temperature
(a) T/Jr (b) 10T/Jr (c) 100T/Jr (d) None of these
The solution to MCQ 1 to 24 is available on the next page.
