What Is Coulomb’s Law? | Electric force formula, direction
Last updated on August 12th, 2021 at 02:58 pm
Coulomb’s law gives the electric force acting between two point charges. A point charge is a pointlike object with a nonzero electric charge. Coulomb’s law says that the force between two charges having magnitudes q1 and q2 separated by a distance r is F = ( k q1 q2) / r2 where k is a constant equal to about 8.99 × 109 N⋅m2/C2 in a vacuum.
Constant k for vacuum is expressed with the equation, k = 1/(4πε0) where ε0 (“epsilon-nought”) is the permittivity of free space and is equal to 8.85× 10-12 F/m (farads per metre). If the point charges aren’t in a vacuum, then ε0 is replaced by the permittivity,ε, of the material between them. Air can be treated as a vacuum when using Coulomb’s law.
The direction of the force on each charge is toward the other one if the charges have opposite signs (opposite charges attract each other). And the direction of the force on each charge is away from each other if the signs of the charges are the same (like charges repel each other).
With SI units, the charges are in coulombs (C) and the distance in meters (m), so the force comes out in newtons (N).
The force in Coulomb’s Law Equation (equation 1 above) is called the electric force, or the Coulomb force. This is also known as electrostatic force (“static” because the force is present whether or not the charges are moving).
Equation of Coulomb’s Law | Formula
The equation or formula of Coulomb’s Law gives the electric force acting between two point charges having magnitudes q1 and q2 separated by a distance r, as per the following equation: Electrostatic force = F = ( k q1 q2) / r2 ………. (1) where k is a constant equal to about 8.99 × 109 N⋅m2/C2 in a vacuum.
Constant k for vacuum is expressed with the equation, k = 1/(4πε0) where ε0 (“epsilon-nought”) is the permittivity of free space and is equal to 8.85× 10-12 F/m (farads per metre). If the point charges aren’t in a vacuum, then ε0 is replaced by the permittivity,ε, of the material between them. Air can be treated as a vacuum when using Coulomb’s law.
Direction of Electric Force
The direction of the electric force exerted on one point charge due to another point charge is always along the line that joins the two point charges.
Remember that, unlike the gravitational force, the electric force can either be attractive or repulsive, depending on the signs of the charges.
The direction of the force on each charge is toward the other one if the charges have opposite signs (opposite charges attract each other). And the direction of the force on each charge is away from each other if the signs of the charges are the same (like charges repel each other).

The similarity between electric force formula and gravity formula
There are some similarities between the electric force formula and the gravity formula. These are as follows:
1] Like gravity, the electric force is an inverse square law force. such that The force is proportional to the inverse square of the separation r between two point charges (F ∝ 1/r^2). That means the strength of the force decreases as the separation increases.
2] The strength of the force is also proportional to the magnitude of each of the two charges (∣q1∣ and ∣q2∣) just as the gravitational force is proportional to the mass of each of two interacting objects.
Sample Numerical problem solved using Coulomb’s Law
1 ] What is the force of attraction between the electron and proton in a hydrogen atom?
Solution:
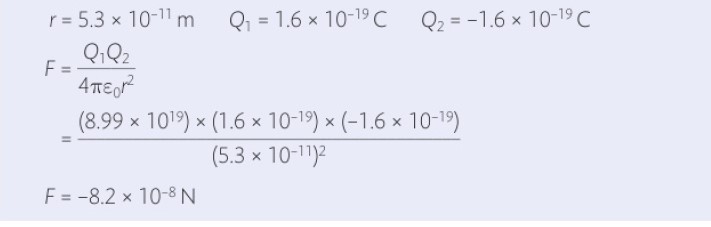
The negative sign here refers to that it’s the force of attraction between electron and proton.
Summary | Take away | Suggested Reading
We studied Coulomb’s Law with its equation or formula and direction. Also solved a sample numerical problem. Now Share this post and make a bookmark in your browser. Now here are a few related posts for your study.
