What is a Symbol equation and how to Balance it?
Reactions such as magnesium burning with oxygen are described with equations. Word equations describe the reactants (the starting substances) and the products (the final substances). Chemical or symbol equations describe what happens to the atoms, and how many of each there are in the reaction.
In this post, we will focus on word equations and chemical equations or symbol equations. We will also see why and how to balance Chemical or symbol equations.
Writing word equation and symbol equation for Burning magnesium & balancing it
When magnesium burns in the air, it reacts with oxygen to form magnesium oxide.
The word equation for this is:
magnesium + oxygen → magnesium oxide
Chemists usually write symbols and formulae rather than names in equations. So, in this word equation, we should write Mg for the element magnesium, O2 for oxygen, and MgO for magnesium oxide, which is composed of Mg2+ and O2- ions.
Mg + O2 → MgO
But, notice that this doesn’t balance. There are two oxygen atoms in O2 on the left and only one oxygen atom in MgO on the right. So, MgO must be doubled to give:
Mg + O2 → 2MgO
Unfortunately, the equation still doesn’t balance. There are now two Mg atoms on the right in 2MgO, but only one on the left in Mg.
This is easily corrected by writing 2Mg on the left to give:
2Mg + O2 → 2MgO
The numbers of different atoms are now the same on both sides of the arrow. This is a balanced chemical equation.
How to Write a balanced equation (steps to follow)
A balanced chemical equation is a summary of the starting substances (reactants) and the products in a chemical reaction, in which the numbers of atoms of each element are the same on both sides of the arrow.
There are three key steps in writing an equation. These can be summarised as follows:
Step 1 – Write a word equation for the reaction:
e.g. hydrogen + oxygen → water
Step 2 – Write symbols for the elements and formulae for the compounds in the word equation:
e.g. H2 + O2 → H2O
Step 3 – Balance the equation by making the number of atoms of each element the same on both sides:
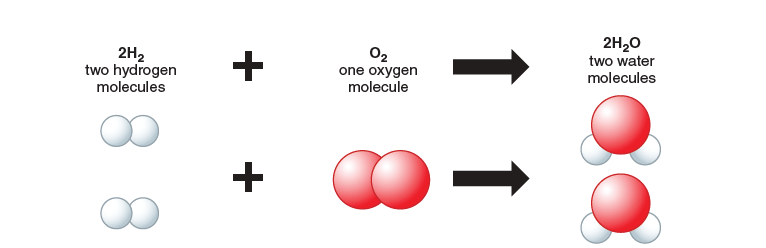
e.g. 2H2 + O2 → 2H2O
Remember that you must never change a formula to make an equation balance.
The formula for water is always H2O and never HO or HO2. Similarly, the formula of magnesium oxide is always MgO.
You can only balance an equation by putting numbers in front of symbols or in front of formulae, i.e. 2Mg and 2H2O.
Why balanced equations are so useful
Balanced chemical equations are more useful than word equations because they show:
- the symbols and formulae of the reactants and products
- the relative numbers of atoms and molecules of the reactants and
products.
Balanced equations also help us to understand how the atoms are rearranged in a reaction.
Showing balanced equations with models
Balanced equations help us to understand how the atoms are rearranged in a reaction. You can see this even better by using models as in Figure 1.
State symbols
State symbols are used in equations to show the state of a substance.
(s) after a formula indicates the substance is a solid. (l) is used for liquid; (g) for gas and (aq) for an aqueous solution (i.e. a substance dissolved in water). For example,
zinc + sulfuric acid → zinc sulfate + hydrogen
Zn(s) + H2SO4(aq) → ZnSO4(aq) + H2(g)
