The van der Waals Equation & Selected van der Waals Constants for Gas Molecules
In this post, we will cover the van der Waals equation and find 2 constants a and b in the equation. We will also find a table full of selected van der Waals Constants for Gas Molecules.
the van der Waals equation
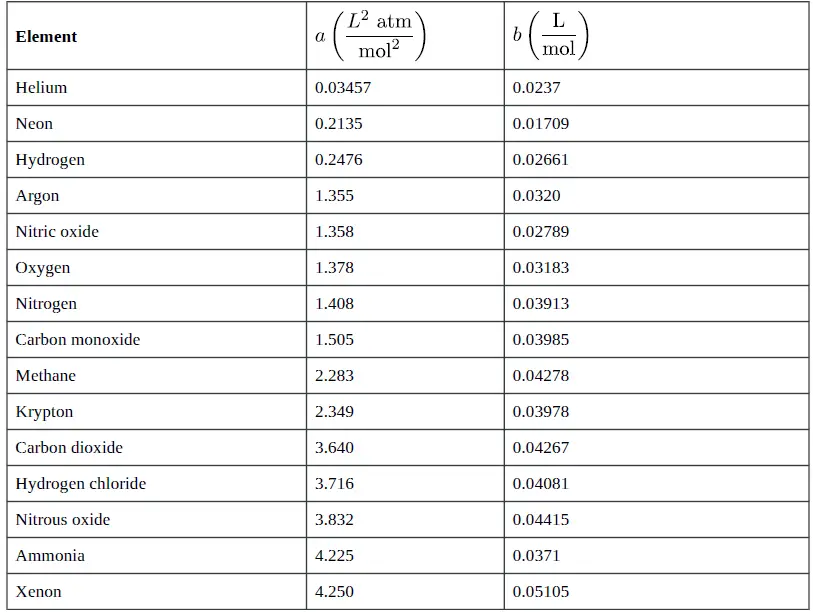
In 1873, Dutch scientist Johannes van der Waals developed an equation that compensates for deviations from ideal gas behavior. The van der Waals equation uses two additional experimentally determined constants: a, which is a term to correct for intermolecular forces, and b, which corrects for the volume of the gas molecules (see Table 1 “Selected van der Waals Constants for Gas Molecules”). The van der Waals equation appears as:
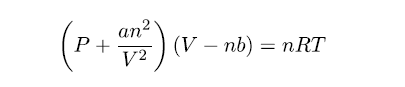
It should be noted that, if the new terms a and b are equal to zero (under ideal conditions), the equation simplifies back to the ideal gas law: PV = nRT.
Table 1: Selected van der Waals Constants for Gas Molecules