Energy Bands in Solids – types of bands, & forbidden energy gap
In an isolated atom, the energy levels of electrons in various orbits can be represented by horizontal lines. Such a diagram is called Energy Level Diagram. Atoms of gases can be considered isolated because of their greater spacing under normal pressure and temperature. Hence the energy levels of such atoms are not affected by other distant atoms. But there are significant changes in the energy levels when atoms exist close together as in solids.
Energy bands in solids & Energy band diagram
There are significant changes in the energy levels when atoms exist close together as in solids. When atoms come closer the outer orbit electrons come close together and even overlap. Because of this interaction, the energy levels are split up. These energy levels are discrete but closely spaced forming almost a continuous range of energy called energy band.
We also know that the number of electrons in the outermost orbit decides valency. These valence electrons are not free to move about from one point to another. If they get sufficient energy they can reach higher energy levels so that they become free to move about. This results in electrical conduction.
Valence Band
The electrons in the outermost orbit of an atom are called valence electrons. The energy band occupied by the valence electrons is called the valence band. The valence band may be either completely filled or partially filled, but can never be empty.
The range of energies possessed by valence electrons is known as the valence band.
Conduction band
The electrons which have left the valence band are called conduction electrons. The range of energy occupied by these electrons is called the conduction band. This band lies next to the valence band. It may be either empty or partially filled with electrons. In the conduction band, electrons move freely and conduct current through the solid.
The range of energies possessed by conducting electrons is known as the conduction band.
If a substance has an empty conduction band, it means that the current conduction is not possible in that substance. Generally, in the insulators conduction band is empty. On the other hand, in conductors, it is partially filled.
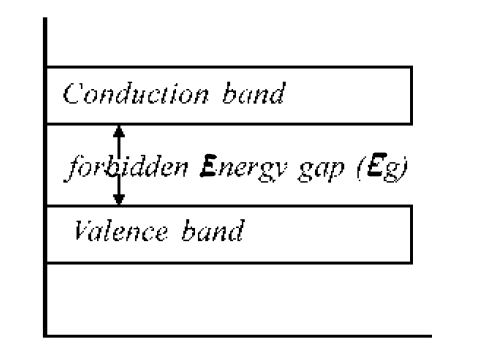
Forbidden Energy Gap
Electrons cannot take Energy Level in between the valence band and conduction band. The separation between the conduction band and valence band on the energy band diagram is known as the forbidden energy gap (band-gap, Eg ).
The width of the energy gap is a measure of the bondage of valence electrons to the atom. The greater the energy gap, the more tightly the valence electrons are bound to the nucleus. In order to push an electron from the valence band to the conduction band, external energy equal to the forbidden energy gap must be supplied.
Classification of solids on the basis of the Forbidden Energy Gap
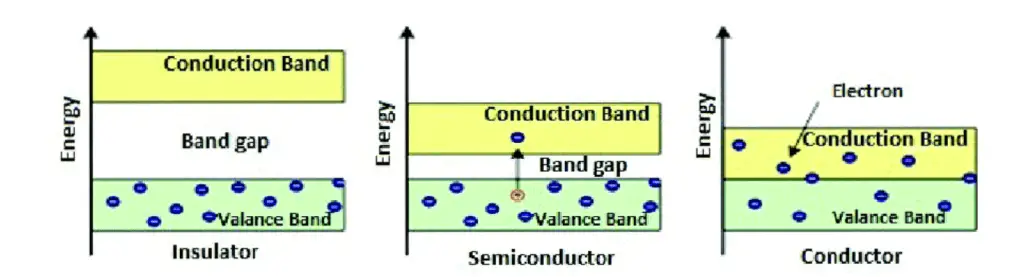
The electrical conduction properties of solids can be explained on the basis of the forbidden energy gap. The solids can be classified into three types. Fig.3 shows the energy band diagram of different solids.
Insulators
As shown in Fig 3 the insulators (eg. Wood, Glass) have a large forbidden energy gap. The electrons are found only in the valence band at room temperature. To achieve conductivity, electrons from the valence band have to move into the conduction band. This is not possible in insulators because of the large forbidden energy gap.
Conductors
Conductors ( eg. Copper, Aluminium) have:
a) overlapping valence and conduction bands
b) plenty of free electrons in the conduction band
In conductors, there is no band gap between the valence band and conduction band. Due to this even, a slight potential difference across the conductor is enough to move the free electrons resulting in electric current.
Semiconductors
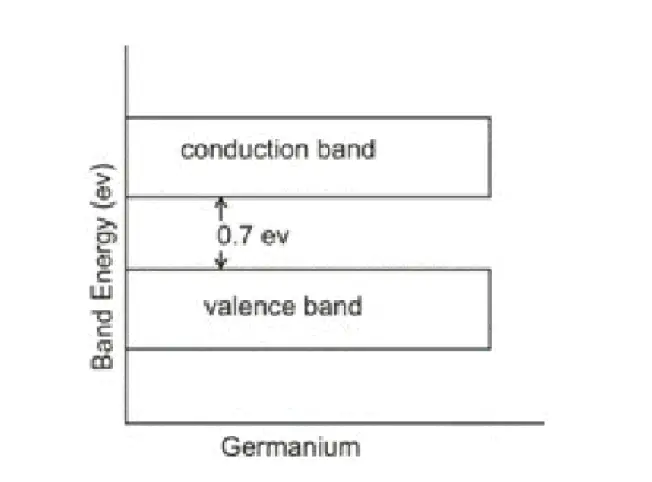
A semiconductor is one whose electrical properties lie in between insulators and conductors (Eg. Silicon and Germanium).
In terms of energy bands, semiconductors at room temperature have a very narrow forbidden energy gap. At 0°K, there are no electrons in the conduction band of semiconductors, whereas their valence band is completely filled.
Therefore at absolute zero temperature, the semiconductor behaves as an insulator.
However, with the increase in temperature, electrons get liberated from the valence band. These free electrons can move towards the conduction band since the energy gap is small.
