Commonly used semiconductors: Germanium & Silicon
Last updated on January 27th, 2022 at 09:34 pm
There are many semiconductors available, but very few of them have practical applications in electronics. The two commonly used materials are Germanium (Ge) and Silicon(Si). In Ge and Si, the energy required to break the covalent bond (energy required to release an electron) is very small. For Ge, it is 0.7 eV and for Si, it is 1.1 eV.
(i) Germanium
Germanium is an earth element and is recovered from the ash of certain coals. Generally recovered Germanium is in the form of germanium dioxide powder which is then purified by chemical reduction techniques.
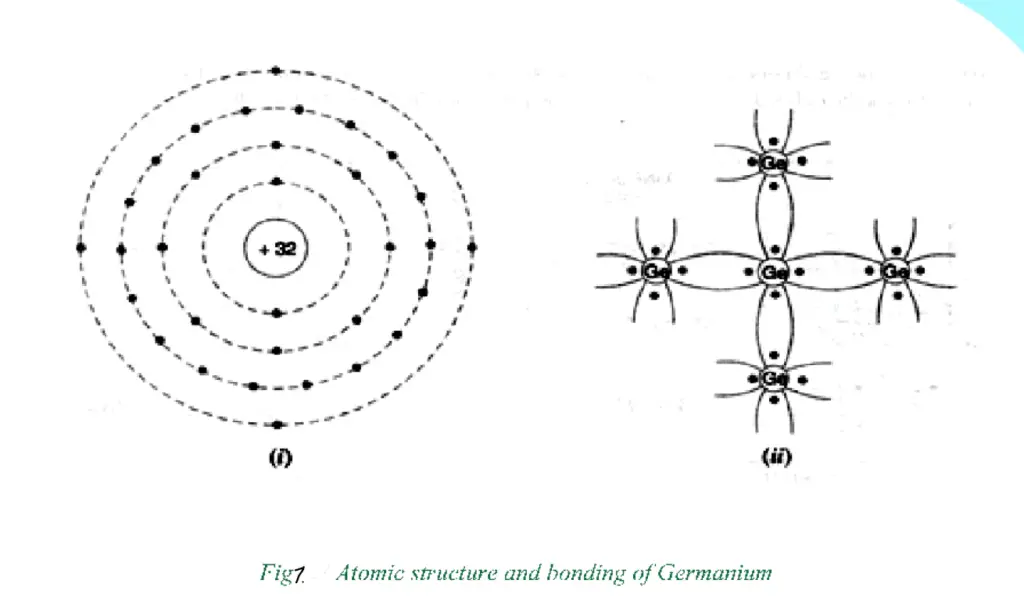
The atomic number of Germanium is 32. Therefore, it has 32 protons and 32 electrons. Two electrons are in the first orbit, eight electrons in the second, eighteen electrons in the third, and four electrons in the outer or valence orbit (See Fig. 1). It is clear that the Germanium atom has four valence electrons making it a tetravalent element.
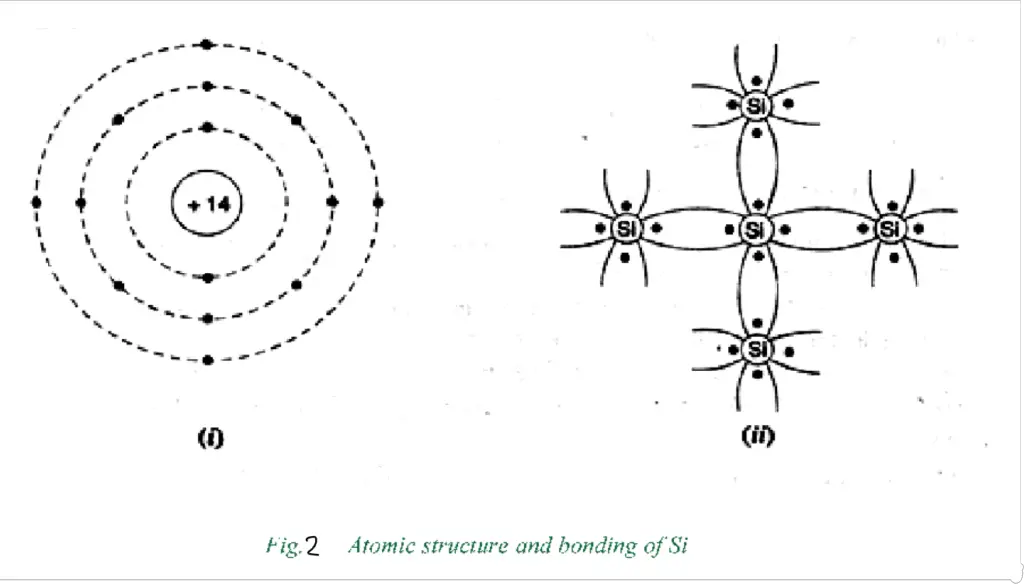
(ii) Silicon
Silicon compounds are commonly available in the form of sand (silicon dioxide). Silicon is obtained by purifying Silicon dioxide by chemical reduction techniques.
The atomic number of silicon is 14. Two electrons are in the first orbit, eight electrons are in the second orbit, and four electrons are in the valence orbit. It is clear that the silicon atom has four valence electrons, that is, it is tetravalent. Fig. 2 shows how various Silicon atoms form covalent bonds.