Different types of Pressure gauges & how they operate
Pressure gauges measure the pressure exerted by a fluid, in other words by a liquid or a gas. We will cover 3 types of pressure gauges here, and these are known as (a) Bourdon gauge (b) U-tube manometer and (c) Mercury barometer. Let’s see how these pressure gauges operate.
Bourdon gauge
In a Bourdon gauge (Figure 1), when fluid pressure is applied, the curved metal tube tries to straighten out and rotates a pointer over a scale.
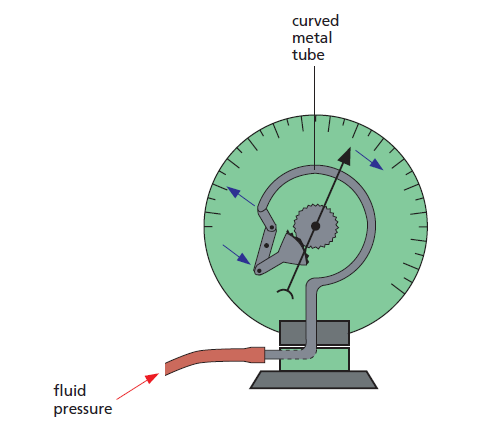
Car oil-pressure gauges and the gauges on gas cylinders are of this type.
U-tube manometer
In Figure 2a each surface of the liquid is acted on equally by atmospheric pressure and the levels are the same.
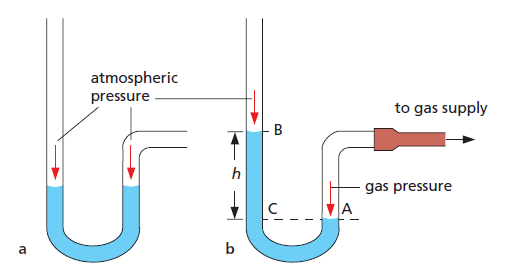
If one side is connected to, for example, the gas supply (Figure 2b), the gas exerts pressure on surface A and level B rises until the pressure of gas = atmospheric pressure + pressure due to liquid column BC.
=> pressure due to liquid column BC = pressure of gas – atmospheric pressure
The pressure of the liquid column BC, therefore, equals the amount by which the gas pressure exceeds atmospheric pressure.
It equals hρg (in Pa) where h is the vertical height of BC (in m) and ρ is the density of the liquid (in kg/m3).
The height h is called the head of liquid and sometimes, instead of stating a pressure in Pa, we say that it is so many cm of water (or mercury for higher pressures).
c) Mercury barometer
A barometer is a manometer that measures atmospheric pressure. A simple barometer is shown in Figure 3.
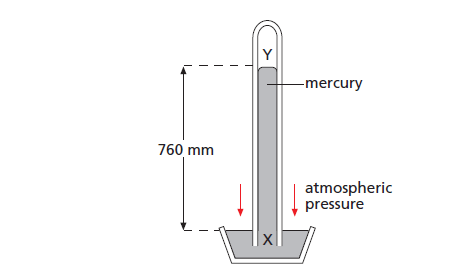
The pressure at X due to the weight of the column of mercury XY equals the atmospheric pressure on the surface of the mercury in the bowl.
The height XY measures the atmospheric pressure in mm of mercury (mmHg).
The vertical height of the column is unchanged if the tube is tilted. The space above the mercury in the tube is a vacuum (except for a little mercury vapour).
