Allotropes of carbon – Diamond & Graphite
Allotropes are different forms of the same element in the same state.
The different forms of solid carbon are called allotropes of carbon. Diamond & Graphite are two crystalline allotropes of carbon. A few other elements also have allotropes. Oxygen has two allotropes – oxygen (O2) and ozone (O3).
Diamond and graphite are both made of pure carbon but these two solids have very different properties and use.
Diamond is hard and clear, whereas graphite is soft and black. Diamonds are used to cut stone and engrave glass, but graphite in pencils is used by artists to achieve a soft, shaded effect on the art paper.
Diamond – an allotrope of carbon | structure of diamond
In diamonds, carbon atoms are joined to each other by strong covalent bonds. Inside the diamond structure (Figure 1), each carbon atom forms covalent bonds with four other carbon atoms. These strong covalent bonds extend through the whole diamond, forming a three-dimensional giant structure.
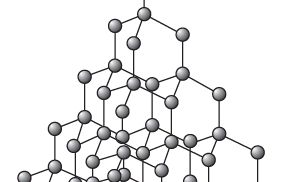
The structure of diamond is described as a giant covalent structure. Every perfect diamond is a giant structure with covalent bonds linking one carbon atom to the next. Only a small number of atoms are shown in the model in Figure 1. In a real diamond, this arrangement of carbon atoms is extended millions and millions of times.
The properties and uses of diamond
1 ) Diamonds are very hard because carbon atoms are linked by very strong covalent bonds. Another reason for its hardness is that the atoms are not arranged in layers so they cannot slide over one another like the atoms in metals.
Most industrial uses of a diamond depend on its hardness.
Diamonds that are not good enough for gems are used in glass cutters and in diamond-studded saws. Powdered diamonds are used as abrasives for smoothing very hard materials.
2) Diamond has a very high melting point. Carbon atoms in diamonds are held in the giant structure by very strong covalent bonds and there are many of them. This means that the atoms cannot vibrate fast enough to break away from their neighbors until very high temperatures are reached. So, the melting point of the diamond is very high.
3 ) Diamond does not conduct electricity. Unlike metals, diamond has no free electrons. All the electrons in the outer shell of each carbon atom are held firmly in covalent bonds. So there are no free electrons in diamond to form an electric current.
Graphite – an allotrope of carbon | structure of Graphite
Figure 2 shows a model of part of the structure of graphite.
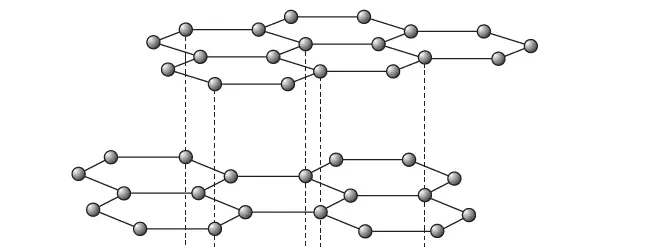
Note that the carbon atoms are arranged in parallel layers in Graphite. Each layer in Graphite contains millions and millions of carbon atoms arranged in hexagons.
Each carbon atom is held strongly in its layer by strong covalent bonds to three other carbon atoms.
So graphite is a giant covalent structure, like a diamond. The distance between neighboring carbon atoms in the same layer is only 0.14 nm, but the distance between the layers is 0.34 nm.
The properties and uses of graphite
1 ) Graphite is a lubricant. In graphite, each carbon atom is linked by strong covalent bonds to three other atoms in its layer. But, the layers are 2½ times further apart than carbon atoms in the same layer. This means that the forces between the layers are weak. If you rub graphite, the layers slide over each other and onto your fingers. This property has led to the use of graphite as the ‘lead’ in pencils and as a lubricant. The layers of graphite slide over each other like a pile of wet microscope slides.
2) Graphite has a high melting point. Although the layers of graphite move over each other easily, it is difficult to break the strong covalent bonds between carbon atoms within one layer. The covalent bonds are strong and there are so many of them. Because of this, graphite does not melt until 3730°C and it does not boil until 4830°C. So, it is used to make crucibles for molten metals. The bonds between carbon atoms in the layers of graphite are so strong that graphite fibers with the layers arranged along the fiber are stronger than steel. These fibers are used to reinforce metals and broken bones. Graphite fibers have been used to reinforce the shaft of badminton rackets.
3 ) Graphite conducts electricity. The forces of attraction between the layers of graphite are fairly weak. The electrons can move along the layers from one atom to the next when graphite is connected to a battery. So graphite will conduct electricity, unlike diamond and other non-metals. Because of this unusual property, graphite is used for electrodes in industry and as the positive terminals in dry cells (batteries).
