Electrolysis & manufacturing of Sodium & chlorine
Sodium and chlorine are manufactured by the electrolysis of molten sodium chloride. When an electric current passes through molten sodium chloride, a shiny bead of sodium is produced at the cathode, and chlorine gas forms at the anode. This decomposition is caused by electrical energy in the current. In this post, we will be explaining the electrolysis of molten sodium chloride.
Explanation of Electrolysis & manufacturing of Sodium & chlorine
Metal/non-metal compounds are composed of ions – positive metal ions and negative non-metal ions.
Sodium chloride is a compound of the metal sodium and the non-metal chlorine and consists of positive sodium ions, Na+, and negative chloride ions, Cl–. Its formula is Na+Cl–, or simply NaCl because the positive charge on one Na+ ion balances the negative charge on one Cl– ion to produce neutral NaCl.
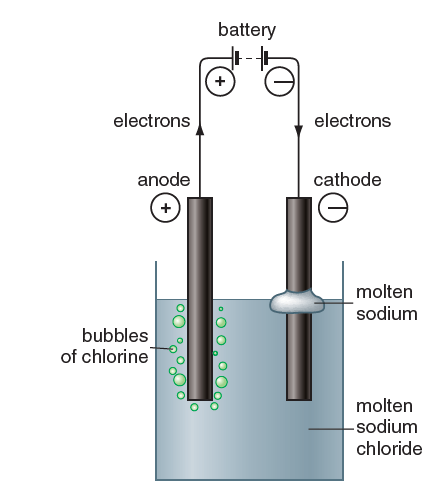
molten sodium chloride
During electrolysis charged ions move towards the electrode of opposite charge.
So, when molten sodium chloride is electrolysed, Na+ ions move towards the negatively charged cathode. Here, they combine with negative electrons on the cathode to form neutral sodium.
Using Half-equation this can be shown as:

The Cl– ions are attracted to the positive anode. here they lose an electron to the anode leaving neutral chlorine atoms.
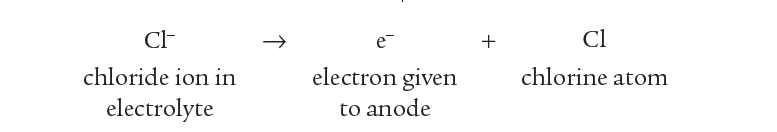
The Cl atoms then join up in pairs to form molecules of chlorine gas, Cl2.

So, overall for the reaction at the cathode where chloride ions become chlorine gas:
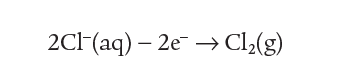
These equations show that during electrolysis Na+ ions take electrons from the cathode and Cl– ions give up electrons to the anode. The electric current is being carried through the molten sodium chloride by ions. The electrolysis of other molten and aqueous substances can also be explained in terms of ions.
The equation summarising the reaction at one electrode is called a half equation because it represents only half of the overall electrolysis.
