Determination of Density of a solid and a liquid
Determination of Density of a Solid by Using a Measuring Cylinder
Dividing the mass of a solid by its volume gives its density. The mass of a solid can be determined accurately by using a physical balance. The volume of an irregular-shaped solid can be determined by using a measuring jar (cylinder). To measure the volume of a solid, note the initial reading in the measuring jar and immerse the solid into the jar. Note down the final reading. The difference between the final and the initial readings gives the volume of the solid. Divide the mass by the volume and find out the density.
Determination of Density of a Liquid by Using a Density Bottle
A density bottle is a specially designed glass bottle available in different capacities. Generally, a density bottle of capacity 50 ml is used for finding the density of various liquids.
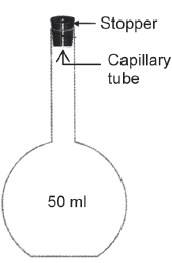
A density bottle is a long-necked bottle provided with a ground glass stopper as shown in Fig 1.
The stopper has a capillary tube. When the bottle is filled with a given liquid and the stopper is inserted, the liquid rises through the capillary tube and overflows. This ensures the accurate volume of a liquid being taken in the density bottle, whenever it is filled with different liquids.
- In order to find the density of a given liquid, the density bottle is washed with the distilled water and dried in hot air.
- The mass of the density bottle with the stopper is determined by using a physical balance. Say, it is Mb
- The density bottle is then filled with distilled water and the stopper is inserted. The water that overflows is wiped and the mass is determined (using a physical balance). Say, this mass is Mb + w
- After determining the mass of the bottle with distilled water, the distilled water is poured out and the bottle is dried in hot air.
- Now the bottle is filled with the liquid and the stopper is inserted.
- The liquid that overflows is wiped out and the mass of the density bottle, filled with the liquid is measured, using a physical balance. Say, this mass is Mb + l
- ** We can find the mass of a liquid and the distilled water, by subtracting the mass of the empty density bottle from the mass of density bottle filled with liquid and distilled water. Mass of liquid Ml = Mb + l – Mb. And, the mass of distilled water Mw = Mb + w – Mb.
- The volume of liquid is equal to the volume of water. (same density bottle is used)
- The density of the liquid can be found as follows:
(first, we have to find the relative density and then calculate the density of the liquid)- Relative density of the liquid =density of the liquid/density of water
= (mass of liquid/volume of liquid)÷(mass of water/volume of water)
= mass of liquid/mass of water [ As, the volume of liquid = volume of water]
= Ml / Mw
As we have found out the values of both Ml and Mw (see above **) already, so we can get the Relative density(RD) of the liquid easily. - Now, as we have determined the relative density of the liquid, now the density of the liquid can be found out easily using the known value of density of water (1 g cm–3 in CGS or 1000 kg m-3 in SI), and using this formula:
The density of the liquid = Relative density of the liquid * density of water
- Relative density of the liquid =density of the liquid/density of water
