Intermolecular Forces – Types & features
The forces of attraction and repulsion between interacting particles (atoms or molecules) are called intermolecular forces. Such forces exist in all states of matter and are responsible for many structural features and physical properties of matter. The intermolecular forces are weak forces. In this post, we will discuss different types of intermolecular forces with their features and characteristics.
Relevant Facts for revision
- Non-polar molecular forces are the weakest forces that form London dispersed forces.
- Molecule is the smallest fundamental unit of a chemical compound that can participate in a chemical reaction. It is made with a number of atoms bound together.
- A polar molecule is one that has one end that is slightly positive and the other end that is slightly negative. Polar molecules are formed where the electronegativity of the bonded atoms differs.
- When electrons are exchanged equally between atoms in a diatomic molecule or when polar bonds in a larger molecule cancel each other out, non-polar molecules form.
van der Waals forces
Attractive intermolecular forces are known as van der Waals forces. These forces are weak forces.
Such forces (van der Waals forces) come into existence due to the interplay of the positive and negative charges between the neighboring atoms of non-polar molecules when they are very close to each other.
van der Waals forces include (or arise due to the following types of interactions)
- Dispersion forces or London forces
- Dipole-dipole forces
- Dipole-induced dipole forces
- Hydrogen bonding
Dispersion forces or London forces
Atoms and non-polar molecules are electrically symmetrical and have no dipole moment because their electronic charge cloud is symmetrically distributed.
But temporary dipole may develop in such atoms and non-polar molecules.
The force of attraction between two temporary dipoles was first proposed by the German physicist Fritz London. That’s why this force of attraction between two temporary dipoles is known as London Force. Another name for this force is dispersion force. These forces are always attractive.
Explain Temporary dipoles & London force with examples
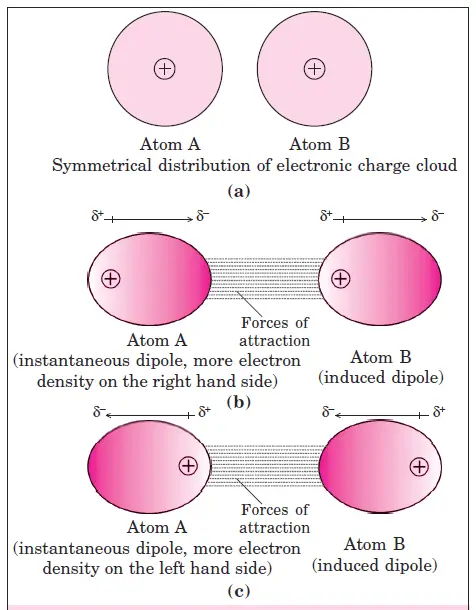
Origin of instantaneous dipole-instantaneous induced dipole (Dispersion forces or London forces)
- In order to understand the origin of these forces, let us consider two noble gas atoms A and B very close to each other. Each atom is uncharged because its electronic charge cloud is symmetrically distributed around the nucleus [Fig. 1 (a)].
- However, due to the motion of the electrons, we can imagine that for a fraction of time, the electronic charge distribution in one of the atoms say ‘A’ becomes unsymmetrical, i.e. charge cloud is more on one side than the other.
- During this very short period, the unsymmetrical centers of positive and negative charges do not coincide. As a result of the instantaneous distribution of electrons in the atom, there is a small temporary dipole known as an instantaneous dipole.
- This instantaneous or transient dipole distorts the electron distribution in the neighboring atom ‘B’ and induces dipole known as instantaneous induced dipole [Fig. 1 (b) and 1 (c)].
- The temporary dipoles of atom ‘A’ and ‘B’ attract each other. These attractive forces are also called instantaneous dipole and instantaneous induced dipole forces. Similarly, temporary dipoles are induced in non–polar molecules also.
Interaction energy due to London forces
These London forces are always attractive and the interaction energy due to these forces is proportional to the sixth power of the distance between two interacting particles (i.e., 1/r6where r is the distance between two particles. These are significant only at short distances (about 500 pm). These also depend upon the polarizability of the atom or molecule.
Polarizability of an atom or a molecule
The polarizability of an atom or a molecule is a measure of the ease with which the electrons and nuclei can be displaced from their average positions. The more easily the electrons and nuclei can be displaced, the greater is the polarizability. The larger the polarizability, the greater is the magnitude of instantaneous dipole and hence the stronger the London forces.
Dipole-dipole (or Keesom) forces
These types of forces occur in molecules that have permanent electric dipoles such as HCl, NH3, H2O, etc. A polar molecule has separate centers of positive and negative charges and the ends of the diploes possess “partial charges”. These charges are shown by the Greek letter delta (δ).
The partial charges are always less than the unit electronic charge (1.6 × 10–19C).
These Dipole-dipole (or Keesom) forces arise due to interactions between oppositely charged ends of the polar molecules. The positive end of one molecule attracts the negative end of the other molecule and vice versa. For example, the dipole-dipole interactions between the hydrogen chloride molecules are shown in Fig. 2.
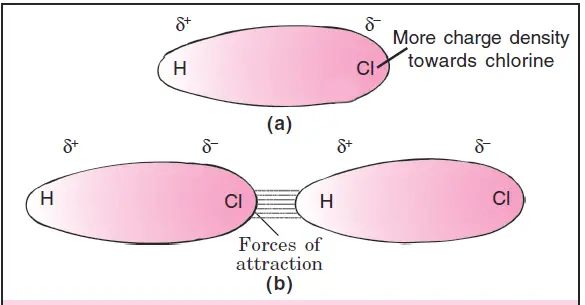
The greater the dipole moment of the molecules, the greater are the forces of attraction. This effect was first studied by Keesom in 1912 and is also referred to as the orientation effect. These forces are also called Keesom forces and produce a net attractive influence.
The interactions due to Dipole-dipole (or Keesom) forces are stronger than London forces. However, these are weaker than ion-ion interactions because only partial charges are involved. The interactions decrease with the increase of distance between the dipoles.
Interaction energy due to Dipole-dipole (or Keesom) forces
As in London forces, the interaction energy for Dipole-dipole (or Keesom) forces is inversely proportional to the distance between polar molecules. The dipole-dipole interaction energy between stationary polar molecules (as in solids) is proportional to 1/r3 and that between rotating polar molecules is proportional to 1/r6, where r is the distance between polar molecules.
Besides dipole-dipole interactions, polar molecules can interact by London forces also
It may be noted that besides dipole-dipole interactions, polar molecules can interact by London forces also. As a result, the cumulative effect is that the total intermolecular forces in polar molecules increase.
Dipole-induced dipole forces
This type of attractive force operates between the polar molecule having a permanent dipole and the molecules having no permanent dipole. For example, if the molecules of a substance are non-polar (such as noble gas, iodine molecule, etc.), they do not possess any dipole. However, when these molecules come into the neighborhood of other molecules which have a permanent dipole, an induced dipole appears on these non-polar molecules.
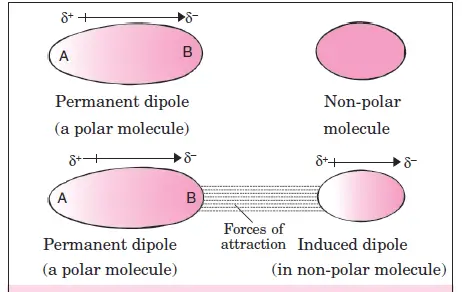
The forces existing between dipole and induced dipole are called dipole-induced dipole forces. The easier the molecules are deformed (easily polarizable), the greater are the attractive forces. This effect was studied by Debye in 1920 and is known as the induction effect.
Interaction energy due to Dipole-induced dipole forces
The interaction energy is also proportional to 1/r6 and depends upon the dipole moment of the polar molecule and the polarizability of the non-polar atom or molecule. Molecules or atoms of larger size have more tendency to get polarized.
cumulative effect of dispersion forces and dipole-induced dipole interactions
In this case, also, the cumulative effect of dispersion forces and dipole-induced dipole interactions exist. In other words, dipole-induced dipole interactions are accompanied by London forces or dispersion forces.
summary ( van der Waals forces )
The above three types of forces (London or dispersion forces, dipole-dipole forces, dipole-induced dipole forces) are collectively known as van der Waals forces. The magnitude of each type of interaction will vary depending upon the type of molecule.
Hydrogen bond
Hydrogen bond is a special case of dipole-dipole interactions.
When a hydrogen atom is bonded to atoms of highly electronegative elements such as fluorine, oxygen, or nitrogen, the hydrogen atom forms a weak bond with the electronegative atom of the other molecule. This weak bond is called the hydrogen bond.
For example, in hydrogen fluoride, HF, hydrogen atom forms a weak bond with fluorine atom of the neighboring molecule, while remaining bonded to its fluorine atom. This may be shown as :
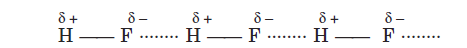
In other words, hydrogen (atom acts as a bridge) between two atoms, holding one atom by a covalent bond and the other atom by a hydrogen bond. The hydrogen bond is represented by the dotted line (….) while the covalent bond is represented by the solid line (—).
As a result of hydrogen bonding, HF exists as a cluster of hydrogen fluoride molecules and is represented as (HF)n. It may be noted that although hydrogen bonding is regarded as being limited to N, O, and F, some molecules containing Cl may also participate in hydrogen bonding.
The energy of the hydrogen bond varies between 10 to 100 kJ mol–1. This is quite a significant amount of energy and therefore, hydrogen bonds are a powerful force in determining the properties of many compounds.
These play a significant role in proteins and nucleic acids.
The strength of the hydrogen bond is determined by the coulombic interactions between the lone pair of electrons of the electronegative atom of one molecule and the hydrogen atom of the other molecule.
The intermolecular forces discussed so far are all attractive forces.
molecules also exert repulsive forces on one another
However, molecules also exert repulsive forces on one another.
When two molecules are brought into close contact with each other, the repulsion forces may also start operating.
These repulsions happen
(a) between the electron clouds of two molecules and
(2) between the nuclei of two molecules.
The magnitude of repulsive forces increases rapidly as the distance between the molecules decreases.
liquids and solids are hard to compress – why?
The magnitude of repulsive forces increases rapidly as the distance between the molecules decreases. This is the reason that liquids and solids are hard to compress. In liquids and solids, the molecules are already close, and therefore, they resist further compression because that would result in the increase of repulsive interactions.
