Static Electricity & Charge – Important Questions and Answers
Last updated on April 12th, 2021 at 08:59 am
This post is going to cover Static electricity, electrostatic charge, and related topics with important questions and answers. This Q&A set will be helpful for students of different boards including IGCSE, GCSE, ICSE, ISC, CBSE, etc.
- Explain Static Electricity with common examples
- State the law of conservation of electric charge | explain briefly
- Normally when objects are charged by rubbing, they hold their charge only for a limited time and eventually, return to the neutral state. Where does the charge go?
- Why do conductors, insulators, and semiconductors behave differently? – explain from the atomic point of view
- How to charge a neutral or uncharged metal rod? | What are the different ways that can charge an uncharged metal rod?
- Define Charging by Induction
- How to charge two metal spheres with opposite charges by Induction charging? – explain step by step process
- How to Charge a single metal sphere by Induction?
- Explain briefly how charging is done by Conduction
- Dangers of static electricity
- Uses of static electricity
- Related Posts | Suggested Posts
Explain Static Electricity with common examples
When a piece of hard rubber, a glass rod, or a plastic ruler rubbed with a cloth then this effect called static electricity is observed. You can readily pick up small pieces of paper with a plastic comb or ruler that you have just vigorously rubbed with even a paper towel. (See Fig. 1)
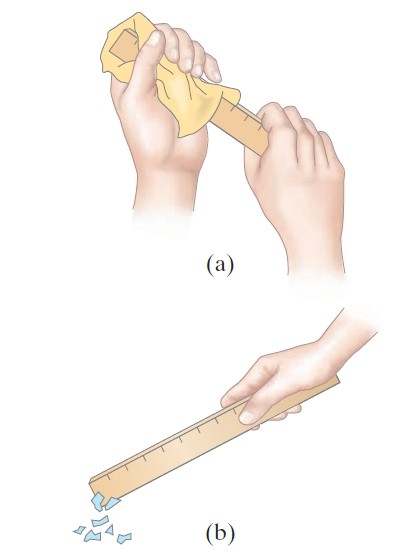
and (b) bring it close to some tiny pieces of paper.
We experience static electricity when combing our hair or when taking a synthetic blouse or shirt from a clothes dryer. And we may feel a shock when we touch a metal doorknob after sliding across a car seat or walking across a synthetic carpet. In each case, an object becomes “charged” as a result of rubbing, and is said to possess a net electric charge.
State the law of conservation of electric charge | explain briefly
The law of conservation of electric charge, which states that the net amount of electric charge produced in any process is zero; i.e., no net electric charge can be created or destroyed.
If one object (or a region of space) acquires a positive charge, then an equal amount of negative charge will be found in neighboring areas or objects. No violations have ever been found, and the law of conservation of electric charge is as firmly established as those for energy and momentum.
Normally when objects are charged by rubbing, they hold their charge only for a limited time and eventually, return to the neutral state. Where does the charge go?
Normally when objects are charged by rubbing, they hold their charge only for a limited time and eventually return to the neutral state. Usually, the excess charge “leaks off” onto water molecules in the air. This is because water molecules are polar—that is, even though they are neutral, their charge is not distributed uniformly. (Fig. 2). [Water molecule has opposite charges on different ends, hence it is called a “polar” molecule.]
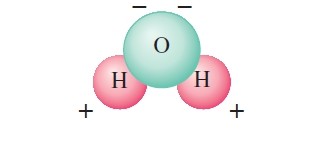
Thus the extra electrons on, say, a charged plastic ruler can “leak off” into the air because they are attracted to the positive end of water molecules.
A positively charged object, on the other hand, can be neutralized by the transfer of loosely held electrons from water molecules in the air.
On dry days, static electricity is much more noticeable since the air contains fewer water molecules to allow leakage of charge. On humid or rainy days, it is difficult to make any object hold a net charge for long.
Why do conductors, insulators, and semiconductors behave differently? – explain from the atomic point of view
Conductors, insulators, and semiconductors behave differently because of either availability or non-availability of free electrons.
From the atomic point of view, the electrons in an insulating material are bound very tightly to the nuclei. In a good metal conductor, on the other hand, some of the electrons are bound very loosely and can move about freely within the metal (although they cannot leave the metal easily) and are often referred to as free electrons or conduction electrons.
When a positively charged object is brought close to or touches a conductor, the free electrons in the conductor are attracted
by this positively charged object and move quickly toward it. If a negatively charged object is brought close to the conductor, the free electrons in the conductor move swiftly away from it.
In a semiconductor, there are many fewer free electrons, and in an insulator, almost none. Hence these behave differently from conductors.
How to charge a neutral or uncharged metal rod? | What are the different ways that can charge an uncharged metal rod?
A neutral or uncharged metal rod can be charged by following any one of the following ways: (1) Charging by Conduction (2) Charging by induction (3) By grounding the metal rod first and then bringing a charged object near the given metal rod and quickly disconnecting the rod from the ground.
Define Charging by Induction
If a charged object is brought near a conducting surface, even without physical contact, electrons will move in the conducting surface. Charging by Induction is the charging of an object without direct contact.
How to charge two metal spheres with opposite charges by Induction charging? – explain step by step process
We will discuss the step by step process of charging by Induction using 2 metal spheres.
In other words, the Induction Charging method will be used to charge two metal spheres with opposite charges. Refer to the following diagram as a supporting figure.
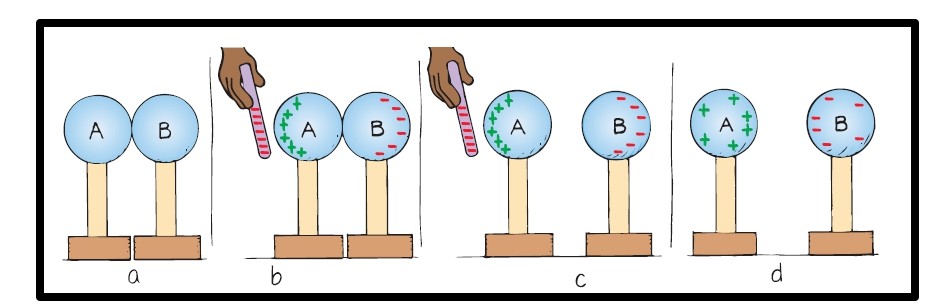
- In Figure a, the uncharged insulated metal spheres touch each other, so in effect, they form a single noncharged conductor.
- In Figure b, a negatively charged rod is held near sphere A. Electrons in the metal are repelled by the rod, and the excess negative charge has moved onto sphere B, leaving sphere A with an excess positive charge. The charge on the two spheres has been redistributed or induced.
- In Figure c, the spheres are separated while the rod is still present.
- In Figure d, the rod has been removed, and the spheres are charged equally and oppositely. They have been charged by induction, which is the charging of an object without direct contact. Since the charged rod never touched them, it retains its initial charge.
How to Charge a single metal sphere by Induction?
In this process, a single metal sphere can be charged by the induction method.
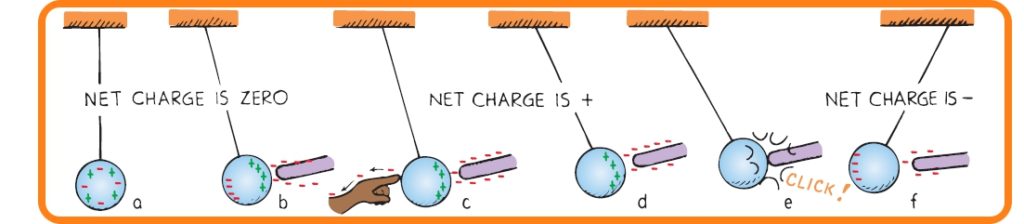
- Consider a metal sphere that hangs from a nonconducting string.
- In Figure (a), the net charge on the metal sphere is zero.
- In Figure (b), a charge redistribution is induced by the presence of the charged rod. The net charge on the sphere is still zero.
- In Figure (c), touching the sphere removes electrons by contact. [When we touch the metal surface with a finger, charges that repel each other have a conducting path to a practically infinite reservoir for the electric charge—the ground. When we allow charges to move off (or onto) a conductor by touching it, it is common to say that we are grounding it.]
- In Figure (d), the sphere is left positively charged. Now if the string is held tightly and the sphere is not allowed to move towards the rod then the positive charge of the sphere can be retained and it can be considered as a positively charged object. But if the sphere is allowed to move then a different scenario appears and that is described in the next 2 points.
- In Figure (e), the sphere is attracted to the negative rod; it swings over to it and touches it. Now electrons move onto the sphere from the rod. The sphere has been negatively charged by contact.
- In Figure (f), the negative sphere is repelled by the negative rod.
Explain briefly how charging is done by Conduction
Suppose a positively charged metal object A is brought close to an uncharged metal object B. If the two objects touch, the free electrons in the neutral one are attracted to the positively charged object and some of those electrons will pass over to it,
(see the Figure below )
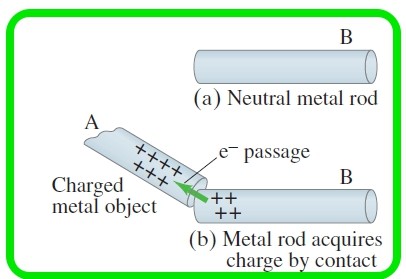
Since object B, originally neutral, is now missing some of its negative electrons, it will have a net positive charge. This process is called charging by conduction, or “by contact,” and the two objects end up with the same sign of charge.
Dangers of static electricity
Here are a few examples of the dangers of static electricity. The dangers are as follows: (1) Lightning – Thunderclouds carry charges (2) Refuelling – Sparks from static electricity can be dangerous when flammable vapor is present (3) Operating theatres as dust and germs are attracted by charged objects (4) Computers are vulnerable to electrostatic damage
Uses of static electricity
a) Flue-ash precipitation
An electrostatic precipitator removes the dust and ash that goes up the chimneys of coal-burning power stations. It consists of a charged fine wire mesh which gives a similar charge to the rising particles of ash. They are then attracted to plates with an opposite charge. These are tapped from time to time to remove the ash, which falls to the bottom of the chimney from where it is removed.
b) Photocopiers
These contain a charged drum and when the paper to be copied is laid on the glass plate, the light reflected from the white parts of the paper causes the charge to disappear from the corresponding parts of the drum opposite. The charge pattern remaining on the drum corresponds to the dark-colored printing on the original. Special toner powder is then dusted over the drum and sticks to
those parts which are still charged. When a sheet of paper passes over the drum, the particles of toner are attracted to it and fused into place by a short burst of heat.
c) Inkjet printers
In an inkjet printer, tiny drops of ink are forced out of a fine nozzle, charged electrostatically, and then passed between two oppositely charged plates; a negatively charged drop will be attracted towards the positive plate causing it to be deflected. The amount of deflection and hence the position at which the ink strikes the page is determined by the charge on the drop and the p.d. between the plates; both of these are controlled by a computer. About 100 precisely located drops are needed to make up an individual letter but very fast printing speeds can be achieved.
